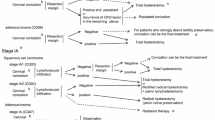
Abstract
The Japan Society of Gynecologic Oncology (JSGO) Guidelines 2017 for the Treatment of Uterine Cervical Cancer are for the purpose of providing standard treatment strategies for cervical cancer, indicating treatment methods currently considered appropriate for cervical cancer, minimizing variances in treatment methods among institutions, improving the safety of treatment and prognosis of diseases, reducing the economic and psychosomatic burden of patients by promoting performance of appropriate treatment, and enhancing mutual understanding between patients and healthcare professionals. The guidelines were prepared through consensus of the JSGO Guideline Committee, based on careful review of evidence gathered through the literature searches and in view of the medical health insurance system and actual clinical practice situations in Japan. The guidelines comprise eight chapters and five algorithms. The main features of the 2017 revision are as follows: (1) evidence was collected using a search formula and with cooperation of the Japan Library Association. The bibliographical search formula was placed at the end of the book; (2) regarding clinical questions (CQs) where evidence or clinical inspection in Japan was lacking, opinions of the Guidelines Committee were described as “proposals for future directions”; (3) cervical intraepithelial neoplasia (CIN) 3 and adenocarcinoma in situ (AIS) were treated as a cervical precancerous lesion; (4) the CQs of endoscopic surgery, radical trachelectomy, and sentinel node biopsy were newly added in Chapter 3, “primary treatment for stage IB–II cervical cancer”; and (5) the CQ about hormone replacement therapy after cancer treatment was newly established. Each recommendation is accompanied by a classification of recommendation categories based on the consensus reached by the Guideline Committee members. Here, we present the English version of the JSGO Guidelines 2017 for the Treatment of Uterine Cervical Cancer.





Similar content being viewed by others
Abbreviations
- AIS:
-
Adenocarcinoma in situ
- CCRT:
-
Concurrent chemoradiotherapy
- CIN:
-
Cervical intraepithelial neoplasia
- COIs:
-
Conflicts of interests
- CQs:
-
Clinical questions
- CTV:
-
Clinical target volume
- EORTC:
-
European Organization for Research and Treatment of Cancer
- FIGO:
-
International Federation of Gynecology and Obstetrics
- Gy:
-
Gray
- HPV:
-
Human papillomavirus
- HRT:
-
Hormone replacement therapy
- HSIL:
-
High-grade squamous intraepithelial lesion
- IMRT:
-
Intensity-modulated radiation therapy
- JCOG:
-
Japan Clinical Oncology Group
- JGOG:
-
Japanese Gynecologic Oncology Group
- JSCO:
-
Japan Society of Clinical Oncology
- JSGO:
-
Japan Society of Gynecologic Oncology
- LSIL:
-
Low-grade squamous intraepithelial lesion
- Minds:
-
Medical Information Network Distribution Service
- NAC:
-
Neoadjuvant chemotherapy
- NCCN:
-
National Comprehensive Cancer Network
- PTV:
-
Planning target volume
- QOL:
-
Quality of life
- RCT:
-
Randomized controlled trial
- RTOG:
-
Radiation Therapy Oncology Group
- SIL:
-
Squamous intraepithelial lesion
- TC:
-
Paclitaxel plus carboplatin
- TP:
-
Paclitaxel plus cisplatin
- WBRT:
-
Whole-brain radiation therapy
- WHO:
-
World Health Organization
- 3D-CRT:
-
3 dimensional-conformal radiotherapy
References
National Cancer Center, Japan (2016) Cancer registry and statistics. Ministry of Health, Labour and Welfare, Tokyo
Nagase S, Inoue Y, Umesaki N et al (2010) Evidence-based guidelines for treatment of cervical cancer in Japan: Japan Society of Gynecologic Oncology (JSGO) 2007 edition. Int J Clin Oncol 15:117–124
Ebina Y, Yaegashi N, Katabuchi H et al (2015) Japan Society of Gynecologic Oncology guidelines 2011 for the treatment of uterine cervical cancer. Int J Clin Oncol 20:240–248
Ariyoshi H (2002) Guideline for correct use of antineoplastic agents (draft). General theory. Gan To Kagaku Ryoho 29:970–977
Ochiai K, Okamoto A, Katsumata N (2002) Guidelines for proper use of antineoplastic agents. Gynecologic cancer. Gan To Kagaku Ryoho 29:1047–1054
Fukui T, Yoshida M, Yamaguchi N et al (2007) Minds handbook on preparation of treatment guidelines 2007. Igaku Shoin, Tokyo
Yamaguchi N, Morizane T, Kojimahara N et al (2014) Minds handbook on preparation of treatment guidelines 2014. Igaku Shoin, Tokyo
Stoler M, Bergeron C, Colgan TJ et al (2014) Squmous cell tumours and precursors. In: Kurman RJ, Carcangiu ML, Herrington CS et al (ed) World Health Organization classification of tumours, 4th edn. Tumours of Female Reproductive Organs. Lyon, IARC Press, Lyon, pp 172–182
Japanese Society of Obstetrics and Gynecology, Japan Society of Pathology (2017) The general rules for clinicaland pathological management of uterine cervical cancer, pathological edition, 4th edn. Kanehara-shuppan, Tokyo
van Meurs H, Visser O, Buist MR et al (2009) Frequency of pelvic lymph node metastases and parametrial involvement in stage IA2 cervical cancer: a population-based study and literature review. Int J Gynecol Cancer 19:21–26
Katabuchi H (2016) Gynecologic cancer comittee report in 2014. Acta Obstet Gynaecol Jpn 68:1117–1160
Cervical Cancer Guideline (Version 1) (2016) NCCN clinical practice guidelines in oncology
Long Y, Yao DS, Pan XW et al (2014) Clinical efficacy and safety of nerve-sparing radical hysterectomy for cervical cancer: a systematic review and meta-analysis. PLoS One 9:e94116
Roh JW, Lee DO, Suh DH et al (2015) Efficacy and oncologic safety of nerve-sparing radical hysterectomy for cervical cancer: a randomized controlled trial. J Gynecol Oncol 26:90–99
Ramirez PT, Jhingran A, Macapinlac HA et al (2011) Laparoscopic extraperitoneal para-aortic lymphadenectomy in locally advanced cervical cancer: a prospective correlation of surgical findings with positron emission tomography/computed tomography findings. Cancer 117:1928–1934
Cosin JA, Fowler JM, Chen MD et al (1998) Pretreatment surgical staging of patients with cervical carcinoma: the case for lymph node debulking. Cancer 82:2241–2248
Brockbank E, Kokka F, Bryant A et al (2013) Pre-treatment surgical para-aortic lymph node assessment in locally advanced cervical cancer. Cochrane Database Syst Rev 2013:CD008217
Sevin BU, Nadji M, Lampe B et al (1995) Prognostic factors of early stage cervical cancer treated by radical hysterectomy. Cancer 76:1978–1986
Lin HH, Cheng WF, Chan KW et al (1996) Risk factors for recurrence in patients with stage IB, IIA, and IIB cervical carcinoma after radical hysterectomy and postoperative pelvic irradiation. Obstet Gynecol 88:274–279
Roman LD, Felix JC, Muderspach LI et al (1998) Influence of quantity of lymph-vascular space invasion on the risk of nodal metastases in women with early-stage squamous cancer of the cervix. Gynecol Oncol 68:220–225
Comerci G, Bolger BS, Flannelly G et al (1998) Prognostic factors in surgically treated stage IB-IIB carcinoma of the cervix with negative lymph nodes. Int J Gynecol Cancer 8:23–26
Kim SM, Choi HS, Byun JS (2000) Overall 5-year survival rate and prognostic factors in patients with stage IB and IIA cervical cancer treated by radical hysterectomy and pelvic lymph node dissection. Int J Gynecol Cancer 10:305–312
Trattner M, Graf AH, Lax S et al (2001) Prognostic factors in surgically treated stage Ib-IIb cervical carcinomas with special emphasis on the importance of tumor volume. Gynecol Oncol 82:11–16
Takeda N, Sakuragi N, Takeda M et al (2002) Multivariate analysis of histopathologic prognostic factors for invasive cervical cancer treated with radical hysterectomy and systematic retroperitoneal lymphadenectomy. Acta Obstet Gynecol Scand 81:1144–1151
Morice P, Piovesan P, Rey A et al (2003) Prognostic value of lymphovascular space invasion determined with hematoxylin-eosin staining in early stage cervical carcinoma: results of a multivariate analysis. Ann Oncol 14:1511–1517
Rutledge TL, Kamelle SA, Tillmanns TD et al (2004) A comparison of stages IB1 and IB2 cervical cancers treated with radical hysterectomy. Is size the real difference? Gynecol Oncol 95:70–76
Chen SW, Liang JA, Yang SN et al (2004) Early stage cervical cancer with negative pelvic lymph nodes: pattern of failure and complication following radical hysterectomy and adjuvant radiotherapy. Eur J Gynaecol Oncol 25:81–86
Delgado G, Bundy B, Zaino R et al (1990) Prospective surgical-pathological study of disease-free interval in patients with stage IB squamous cell carcinoma of the cervix: a Gynecologic Oncology Group study. Gynecol Oncol 38:352–357
Lai CH, Hong JH, Hsueh S et al (1999) Preoperative prognostic variables and the impact of postoperative adjuvant therapy on the outcomes of Stage IB or II cervical carcinoma patients with or without pelvic lymph node metastases: an analysis of 891 cases. Cancer 85:1537–1546
Japanese Society of Obstetrics and Gynecology, Japan Society of Pathology (2012) The general rules for clinical and pathological management of uterine cervical cancer, 3rd edn. Kanehara-shuppan, Tokyo
Japanese Society for Radiation Oncology (2012) Guidelines of radiotherapy treatment planning 2012. Kanehara-shuppan, Tokyo
Small W Jr., Mell LK, Anderson P et al (2008) Consensus guidelines for delineation of clinical target volume for intensity-modulated pelvic radiotherapy in postoperative treatment of endometrial and cervical cancer. Int J Radiat Oncol Biol Phys 71:428–434
Japan Clinical Oncology Group, Toita T, Ohno T et al (2010) A consensus-based guideline defining the clinical target volume for pelvic lymph nodes in external beam radiotherapy for uterine cervical cancer. Jpn J Clin Oncol 40: 456–463
Perez CA, Kavanagh BD (2008) Uterine cervix. In: Perez CA, Brady LW (eds) Principles and practice of radiation oncology, 5th edn. Lippincott Williams & Wilkins, Philadelphia, pp 1800–1915
Mundt AJ, Mell LK, Roeske JC (2003) Preliminary analysis of chronic gastrointestinal toxicity in gynecology patients treated with intensity-modulated whole pelvic radiation therapy. Int J Radiat Oncol Biol Phys 56:1354–1360
Jhingran A (2006) Potential advantages of intensity-modulated radiation therapy in gynecologic malignancies. Semin Radiat Oncol 16:144–151
Chen MF, Tseng CJ, Tseng CC et al (2007) Clinical outcome in posthysterectomy cervical cancer patients treated with concurrent Cisplatin and intensity-modulated pelvic radiotherapy: comparison with conventional radiotherapy. Int J Radiat Oncol Biol Phys 67:1438–1444
Klopp AH, Moughan J, Portelance L et al (2016) Pilot study of a computed tomography-compatible shielded intracavitary brachytherapy applicator for treatment of cervical cancer. Pract Radiat Oncol 3:115–123
Murakami N, Okamoto H, Kasamatsu T et al (2014) A dosimetric analysis of intensity-modulated radiation therapy with bone marrow sparing for cervical cancer. Anticancer Res 34:5091–5098
Rotman M, Pajak TF, Choi K et al (1995) Prophylactic extended-field irradiation of para-aortic lymph nodes in stages IIB and bulky IB and IIA cervical carcinomas. Ten-year treatment results of RTOG 79 – 20. JAMA 274:387–393
Haie C, Pejovic MH, Gerbaulet A et al (1988) Is prophylactic para-aortic irradiation worthwhile in the treatment of advanced cervical carcinoma? Results of a controlled clinical trial of the EORTC radiotherapy group. Radiother Oncol 11:101–112
Toita T, Kitagawa R, Hamano T et al (2012) Phase II study of concurrent chemoradiotherapy with high-dose-rate intracavitary brachytherapy in patients with locally advanced uterine cervical cancer: efficacy and toxicity of a low cumulative radiation dose schedule. Gynecol Oncol 126:211–216
Kato S, Ohno T, Thephamongkhol K et al (2010) Multi-institutional phase II clinical study of concurrent chemoradiotherapy for locally advanced cervical cancer in East and Southeast Asia. Int J Radiat Oncol Biol Phys 77:751–757
Toita T, Moromizato H, Ogawa K et al (2005) Concurrent chemoradiotherapy using high-dose-rate intracavitary brachytherapy for uterine cervical cancer. Gynecol Oncol 96:665–670
Sakata K, Sakurai H, Suzuki et al (2008) Results of concomitant chemoradiation for cervical cancer using high dose rate intracavitary brachytherapy: study of JROSG (Japan Radiation Oncology Study Group). Acta Oncol 47:434–441
Monk BJ, Sill MW, McMeekin DS et al (2009) Phase III trial of four cisplatin-containing doublet combinations in stage IVB, recurrent, or persistent cervical carcinoma: a Gynecologic Oncology Group study. J Clin Oncol 27:4649–4655
Moore KN, Herzog TJ, Lewin S et al (2007) A comparison of cisplatin/paclitaxel and carboplatin/paclitaxel in stage IVB, recurrent or persistent cervical cancer. Gynecol Oncol 105:299–303
Kitagawa R, Katsumata N, Shibata T et al (2015) Paclitaxel plus carboplatin versus paclitaxel plus cisplatin in metastatic or recurrent cervical cancer: the open-label randomized phase III trial JCOG0505. J Clin Oncol 33:2129–2135
Grigsby PW (2005) Radiotherapy for pelvic recurrence after radical hysterectomy for cervical cancer. Radiat Med 23:327–330
Thomas GM, Dembo AJ, Myhr T et al (1993) Long-term results of concurrent radiation and chemotherapy for carcinoma of the cervix recurrent after surgery. Int J Gynecol Cancer 3:193–198
Ito H, Shigematsu N, Kawada T et al (1997) Radiotherapy for centrally recurrent cervical cancer of the vaginal stump following hysterectomy. Gynecol Oncol 67:154–161
Noda K, Ikeda M, Yakushiji M et al (1992) A phase II clinical study of cis-diammine glycolato platinum, 254-S, for cervical cancer of the uterus]. Gan To Kagaku Ryoho 19:885–892
Kato T, Nishimura H, Yakushiji M et al (1992) Phase II study of 254-S (cis-diammine glycolato platinum) for gynecological cancer. Gan To Kagaku Ryoho 19:695–701
Tsuda H, Hashiguchi Y, Nishimura S et al (2004) Phase I-II study of irinotecan (CPT-11) plus nedaplatin (254-S) with recombinant human granulocyte colony-stimulating factor support in patients with advanced or recurrent cervical cancer. Br J Cancer 91:1032–1037
Takekuma M, Hirashima Y, Ito K et al (2012) Phase II trial of paclitaxel and nedaplatin in patients with advanced/recurrent uterine cervical cancer: a Kansai Clinical Oncology Group study. Gynecol Oncol 126:341–345
Donegan WL (1983) Cancer and pregnancy. CA Cancer J Clin 33:194–214
Abe Y, Ito K, Okamura C et al (2004) Cervical cytologic examination during physical checkup of pregnant women: cervical cancer screening in women under the age of thirty. Tohoku J Exp Med 204:221–228
Zemlickis D, Lishner M, Degendorfer P et al (1991) Maternal and fetal outcome after invasive cervical cancer in pregnancy. J Clin Oncol 9:1956–1961
Morice P, Uzan C, Gouy S et al (2012) Gynaecological cancers in pregnancy. Lancet 379:558–569
Lanceley A, Fiander A, McCormack et al (2013) Followup protocols for women with cervical cancer after primary treatment. Cochrane Database Syst Rev 2013:CD008767
Elit L, Fyles AW, Devries MC et al (2009) Follow-up for women after treatment for cervical cancer: a systematic review. Gynecol Oncol 114:528–535
Salani R, Backes FJ, Fung MF et al (2011) Posttreatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: Society of Gynecologic Oncologists recommendations. Am J Obstet Gynecol 204:466–478
Matsuura Y, Kawagoe T, Toki N et al (2006) Long-standing complications after treatment for cancer of the uterine cervix–clinical significance of medical examination at 5 years after treatment. Int J Gynecol Cancer 16:294–297
Greimel ER, Winter R, Kapp KS et al (2009) Quality of life and sexual functioning after cervical cancer treatment: a long-term follow-up study. Psychooncology 18:476–482
Park SY, Bae DS, Nam JH et al (2007) Quality of life and sexual problems in disease-free survivors of cervical cancer compared with the general population. Cancer 110:2716–2725
Acknowledgements
We thank the Japanese Society of Obstetrics and Gynecology (JSOG), the Japan Society of Clinical Oncology (JSCO), the Japan Society of Gynecologic and Obstetric Endoscopy and Minimally Invasive Therapy, the Japanese Gynecologic Oncology Group (JGOG), the Japan Association of Obstetricians and Gynecologists (JAOG), the Japan Society of Pathology (JSP), the Japanese Society for Radiation Oncology (JASTRO) for their comments and contributions throughout the project. We also acknowledge the clerical and technical assistance by Ms Toshie Yasuda of JSGO and Ms Yuriko Adachi of Kanehara CO., LTD.
Members of Guidelines Committee (alphabetical order)
Masanori Kaneuchi, Hidetaka Katabuchi, Mikio Mikami, Satoru Nagase, Tsutomu Tabata. Yasuhiro Udagawa, Nobuo Yaegashi.
Members of guidelines drafting committee (alphabetical order)
Tsukasa Baba, Yasuhiko Ebina, Takayuki Enomoto, Kazuhiko Hayashi, Yasuyuki Hirashima, Takashi Iwata, Shingo Kato, Noriyuki Katsumata, Atsumi Kojima, Tetsuji Kurokawa, Masaki Mandai, Yoshiki Mikami, Toshinari Muramatsu, Yutaka Nagai, Hidekatsu Nakai, Kentaro Nakayama, Hitoshi Niikura, Katsutoshi Oda, Hideyuki Ohtake, Masayuki Sekine, Muneaki Shimada, Tsutomu Tabata, Kiyomi Takaishi, Kiyoshi Takamatsu, Kazuhiro Takehara, Hironori Tashiro, Takafumi Toita, Satoshi Yamaguchi, Masanori Yasuda, Kiyoshi Yoshino.
Members of guidelines evaluation committee (alphabetical order)
Daisuke Aoki, Yoichi Aoki, Takuma Fujii, Keiichi Fujiwara, Toru Hachisuga, Toshio Hamatani, Kenichi Hrano, Kazuhiko Ino, Atsuko Itakura, Kiyoshi Ito, Osamu Iwanari, Seiryu Kamoi, Takahiro Kasamatsu, Naoki Kawamura, Hiroaki Kobayashi, Hiroshi Kobayashi, Takeshi Kodaira, Kaneyuki Kubushiro, Etsuko Miyagi, Mika Mizuno, Kenichiro Morishige, Tetsuro Nagasaka, Toshiaki Nakamura, Masahide Ohmichi, Aikou Okamoto, Takashi Onda, Tsuyoshi Saito, Toshiaki Saito, Noriaki Sakuragi, Toyomi Sato, Tadashi Shiraishi, Toru Sugiyama, Nao Suzuki, Mitsuaki Suzuki, Masashi Takano, Nobuhiro Takeshima, Takashi Uno, Kimio Ushijima, Yoh Watanabe, Hiroko Yokota, Hiroyuki Yoshikawa.
Patient advocacy group “Katorea no Mori”.
Funding
Preparation of these guidelines was funded by the JSGO. No financial support was received from any other organization or corporation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
(1) The Conflicts of Interest Committee of the JSGO discloses information on real or potential COIs of the members of the Drafting and Evaluation Committees for the Guidelines, as well as other relevant parties. The information can be confirmed via the Policy on Conflicts of Interest among Clinical Cancer Researchers, which can be viewed at http://www.jsgo.or.jp/topics/index01.html (prepared by JSGO; in Japanese). The following corporations were disclosed by self‑declaration of the Drafting Committee members. ASKA Pharmaceutical Co., Ltd.; Otsuka Pharmaceutical Co., Ltd.; TSUMURA & Co.; Zeria Pharmaceutical Co., Ltd.; Chugai Pharmaceutical Co., Ltd.; Bayer Yakuhin Ltd.; Pfizer Japan Inc.; and Roche Diagnostics Co., Ltd. (2) Each recommendation was determined through consensus of all committee members and was not influenced by the interests of specific organizations, products, or technologies.
Additional information
This article originally appeared in Japanese as Shikyu keigan chiryo gaidorain 2017 nen ban, published by Kanehara, Tokyo, 2017.
About this article
Cite this article
Ebina, Y., Mikami, M., Nagase, S. et al. Japan Society of Gynecologic Oncology guidelines 2017 for the treatment of uterine cervical cancer. Int J Clin Oncol 24, 1–19 (2019). https://doi.org/10.1007/s10147-018-1351-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-018-1351-y